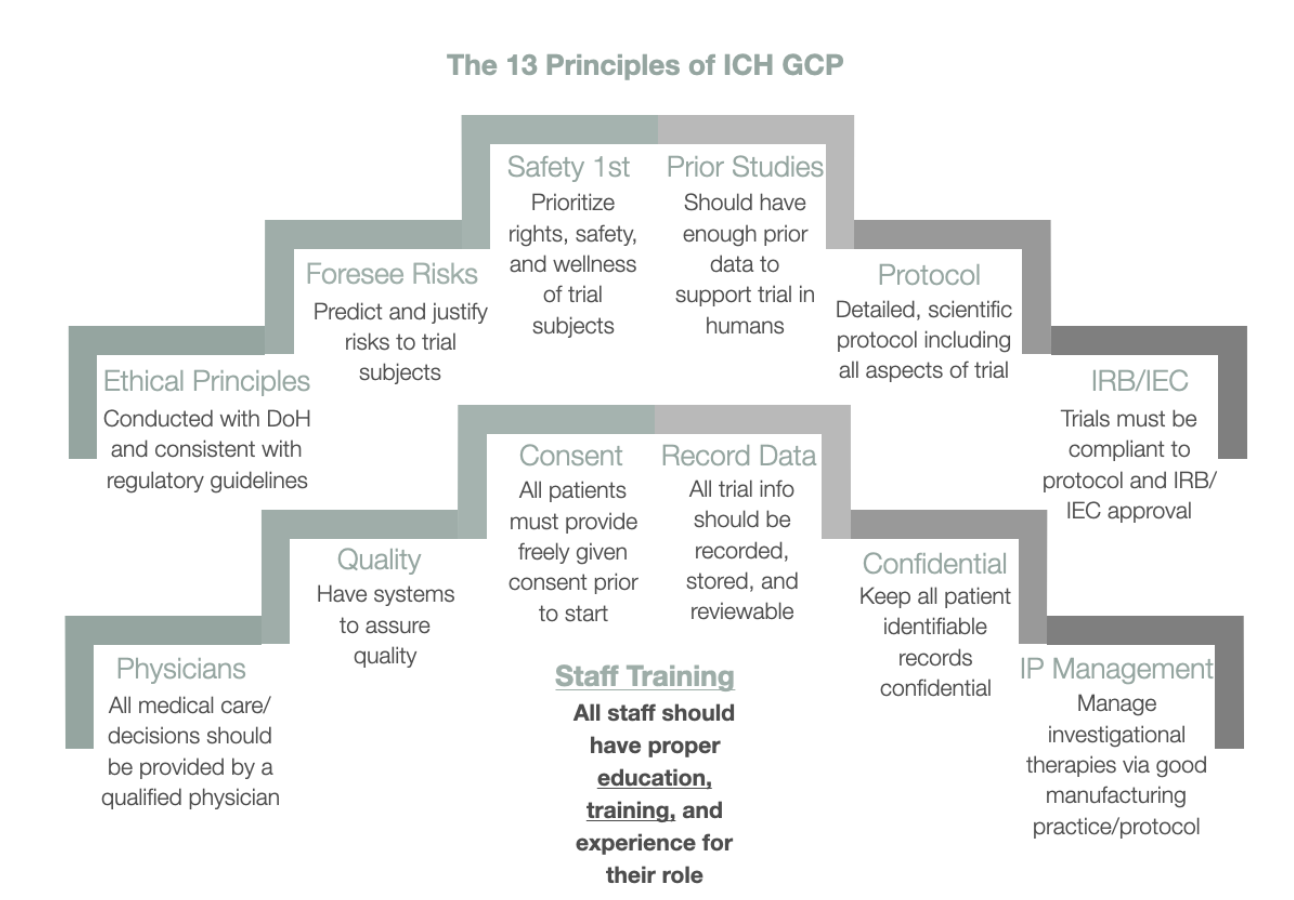
A GCP Primer: Understanding the Basic Application of Good Clinical Practice in the Regulated Environment for the Quality Professional | IVT - GCP

Clinical trials with Medical Devices. Good Clinical Practice lecture by Clinical Director — Pharmahungary

ICH Good Clinical Practice (GCP) E6 (R2) and regulatory requirements for Clinical Trials (Curtin University) - RETProgram

The Good Clinical Practice (GCP) and the responsibilities of pharma sponsors - Avantyo article in Viata Medicala magazine · News · Avantyo

The Good Clinical Practice guideline and its interpretation – perceptions of clinical trial teams in sub‐Saharan Africa - Vischer - 2016 - Tropical Medicine & International Health - Wiley Online Library
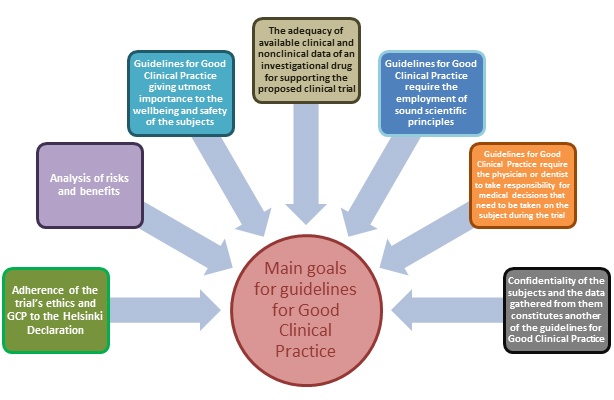
The Good Clinical Practice (GCP) and the responsibilities of pharma sponsors - Avantyo article in Viata Medicala magazine · News · Avantyo
![PDF] The importance of Good Clinical Practice guidelines and its role in clinical trials | Semantic Scholar PDF] The importance of Good Clinical Practice guidelines and its role in clinical trials | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7a90efbaf9d59ee20353e700230ccbf1660f16b5/3-Table2-1.png)

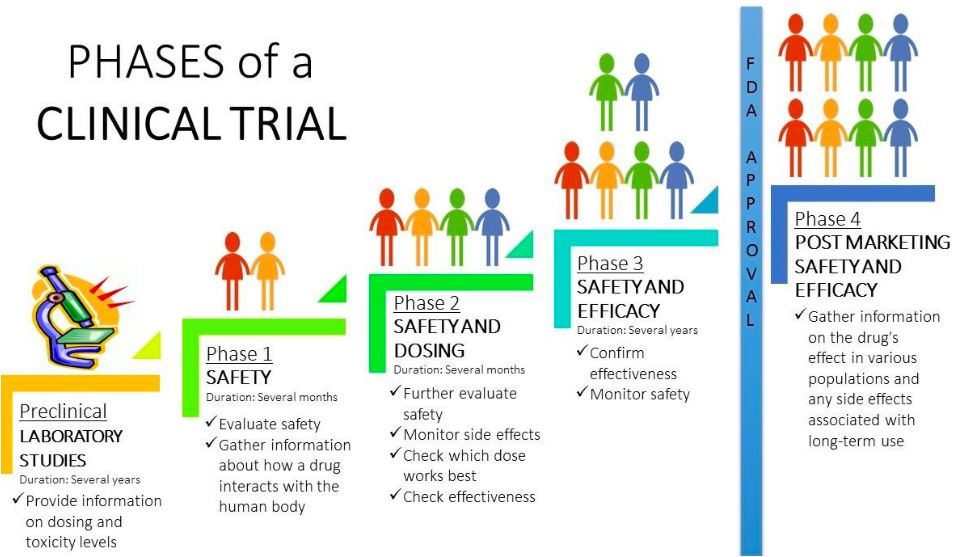

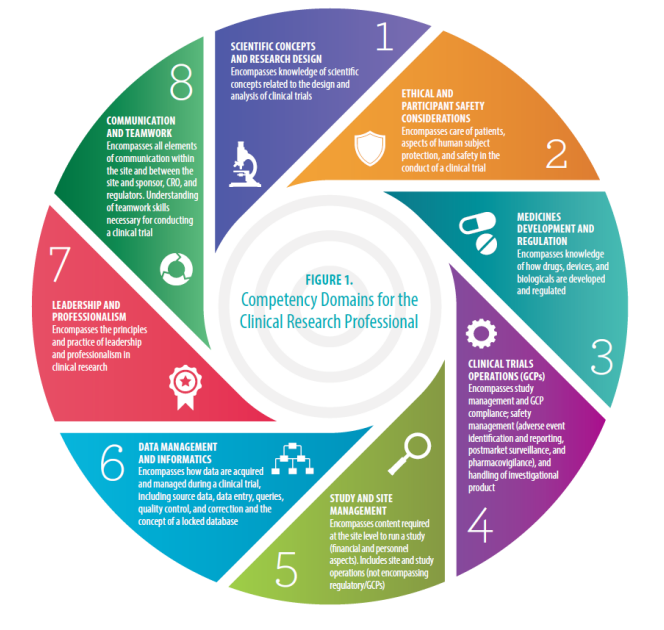





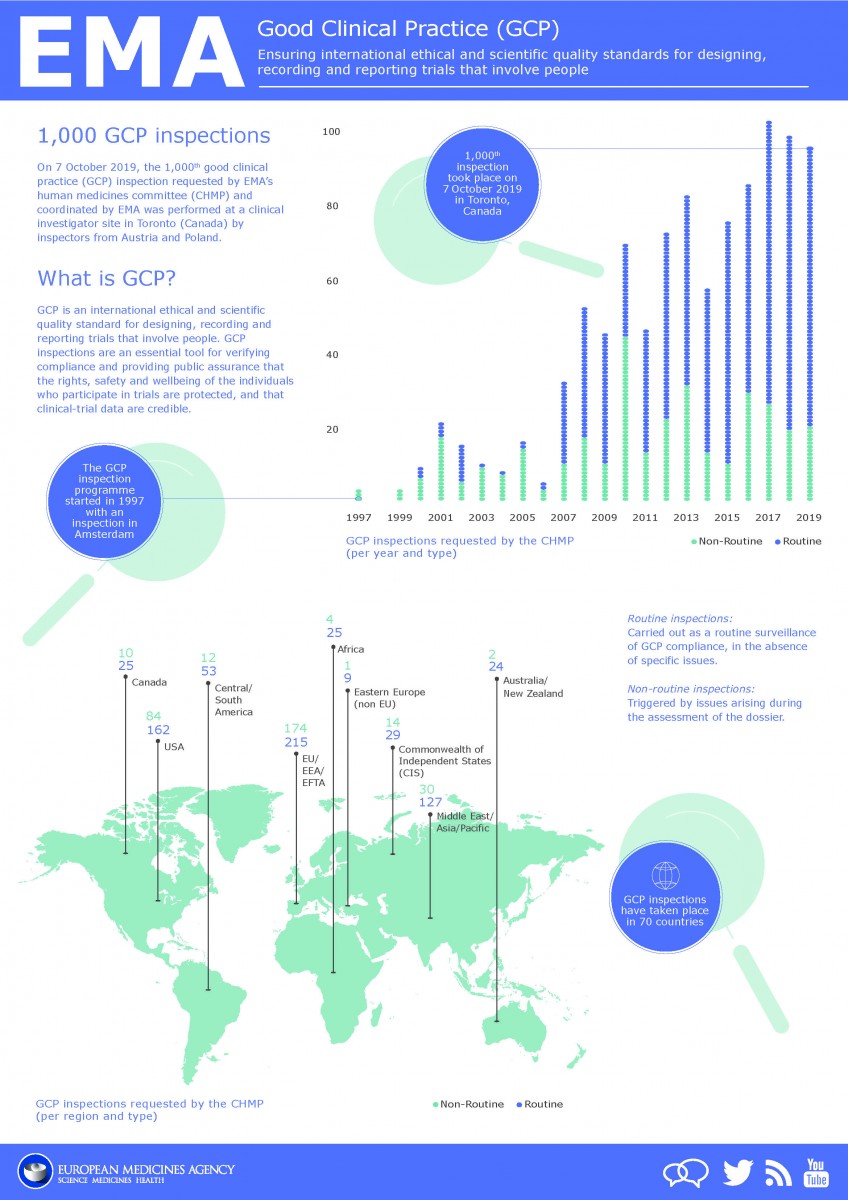

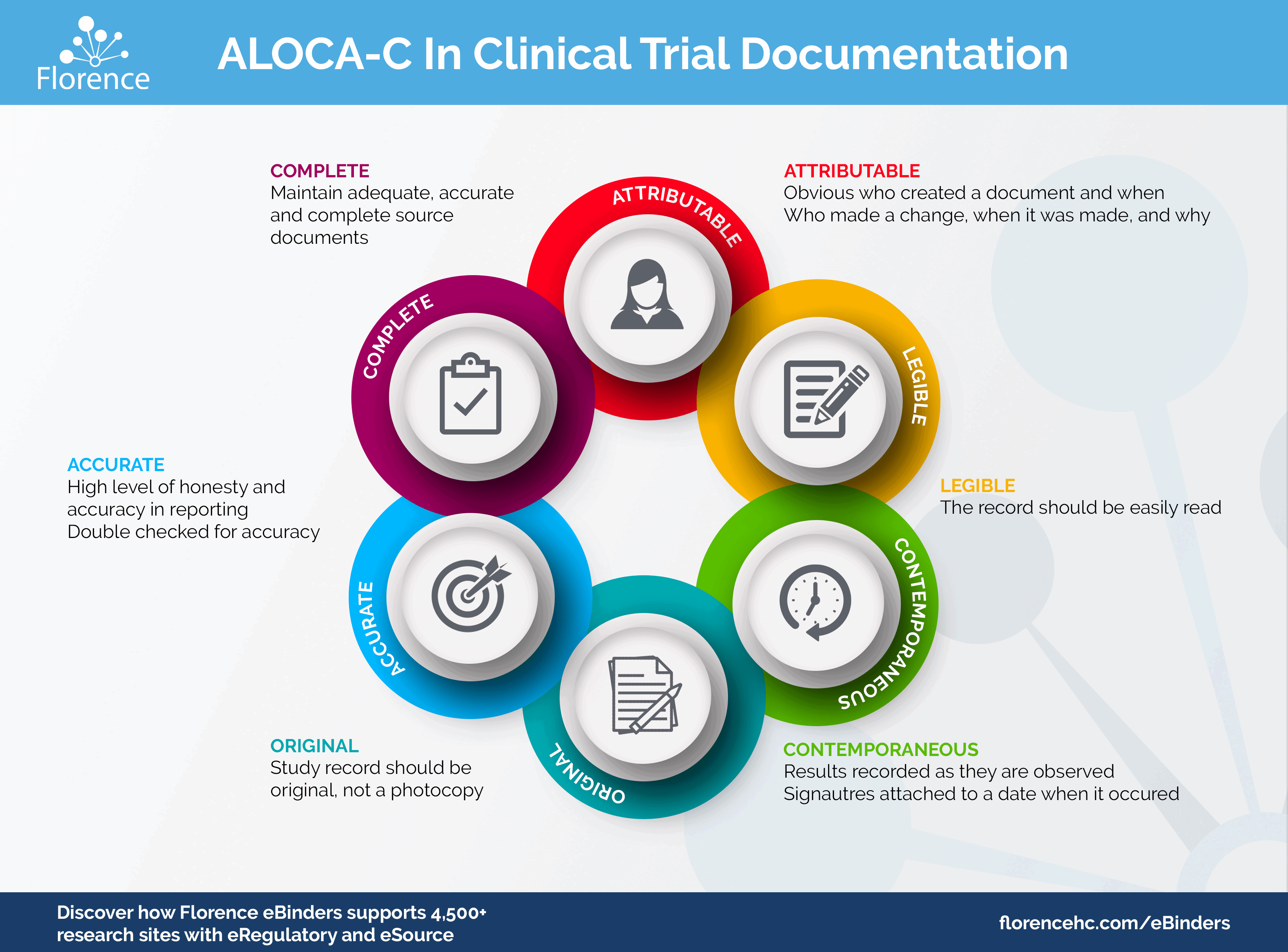
(135).jpg)